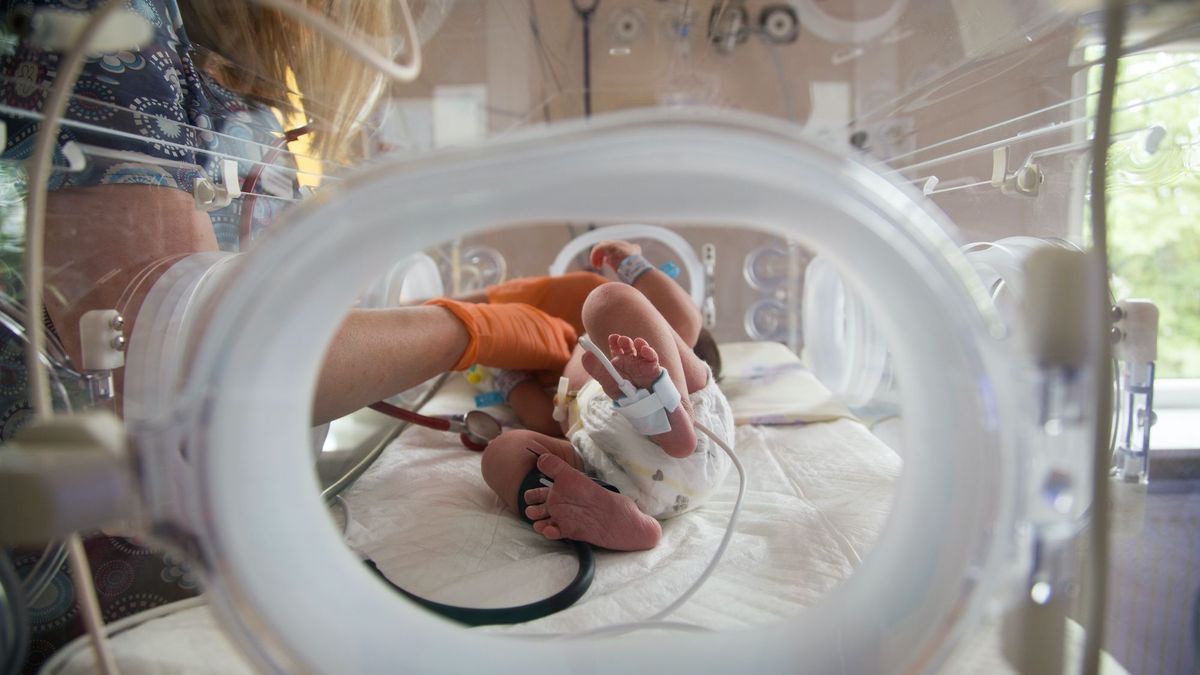
In the United States, ethical and medical discussions began on Wednesday around a delicate theme: can and should we use artificial wombs to save babies born too prematurely? The conditions for a first trial in humans were discussed.
It’s no longer science fiction. Artificial wombs as designed and tested so far on animal models could well be the next medical answer to save very premature babies. In the United States, the health agency, the Food and Drug Administrationheld a meeting Wednesday to discuss their use and the feasibility of a human trial.
What is an artificial uterus?
As one can imagine, an artificial uterus is an artificially created medical environment that mimics the uterus, in the form of a fluid-filled container with lines and tubes attached to babies’ blood vessels, similar to an umbilical cord , with the aim of providing nutrients and medicines.
Unlike incubators already used for premature babies, which are air-filled “modules” that can be heated to help the baby regulate its temperature, these artificial wombs aim to simulate exactly the same environment as a womb. The idea being considered is that a baby born at less than 28 weeks gestation could be placed in the device so that they can grow and develop normally, as if they were still in utero.
For 6 years, this technology has shown its effectiveness on animal models, particularly lambs.
Discussions remain necessary before a human trial
The artificial uterus has been known to scientists for several years, and tested since 2017. During a two-day meeting focused on artificial uterus technology, FDA advisers discussed what the artificial uterus could look like. human trials and complex medical considerations that should be taken into account.
Experts say the development of artificial womb technology could be key to helping save the lives of some babies born prematurely, but unsurprisingly, adapting this technology to humans raises many ethical questions:
- Firstly, the FDA mentions that doctors have a responsibility to “do no harm”. Does this definition apply to artificial incubation? ;
- Other unknowns must also be evaluated: “Whether it will be painful and whether the benefits outweigh the potential risks raises many concerns” pursues the organization;
- Advisors also reviewed potential clinical considerations to fairly assess whether the new technology would be an advancement over currently available care;
- Finally, there are potential long-term consequences of an artificial uterus that remain to be studied, such as potential long-term complications if a uterus is made from materials such as plastic.
The hope of saving many babies in the near future?
The fact that discussions have been initiated, however, remains a strong sign which makes such a device plausible. For the rare teams that have conducted experiments on animal models (like the Children’s Hospital of Philadelphia (CHOP), which has kept 300 prematurely born lambs alive since 2017), approval of the first human clinical trials would be a big step forward. “If this is as successful as we think, ultimately the majority of predicted pregnancies at risk of extreme prematurity would be delivered early in our system, rather than being delivered prematurely on a ventilator.” said Alan Flake, fetal surgeon at CHOP.
To this day, premature birth remains the leading cause of death and disability worldwide among children under the age of five, according to the World Health Organization. In 2020, there were approximately 13.4 million premature births worldwide.
