The list of products that pharmacists can sell is regularly updated. In a new decree dated September 1, 2023, “injectable devices based on hyaluronic acid” and “products not having a medical purpose based on injectable hyaluronic acid” have just been added.
A new decree has just been published on September 1, 2023, concerning wrinkle filler products based on hyaluronic acid, to adapt French law to European law. This decree corrects the impact of a European text, in order to allow the sale of these products in pharmacies. And this, despite the risks of clandestine injections already mentioned many times…
Increased risks of misuse and illegal practice of medicine
The published text therefore authorizes the sale in pharmacies of “hyaluronic acid injectable devices” and of “products not intended for medical use based on injectable hyaluronic acid“.
This easier access makes health authorities fear an increased risk of misuse and illegal practice of these injections, particularly for aesthetic purposes. Injections which, remember, should only be carried out by doctors, as the National Medicines Safety Agency (ANSM) points out on its site.
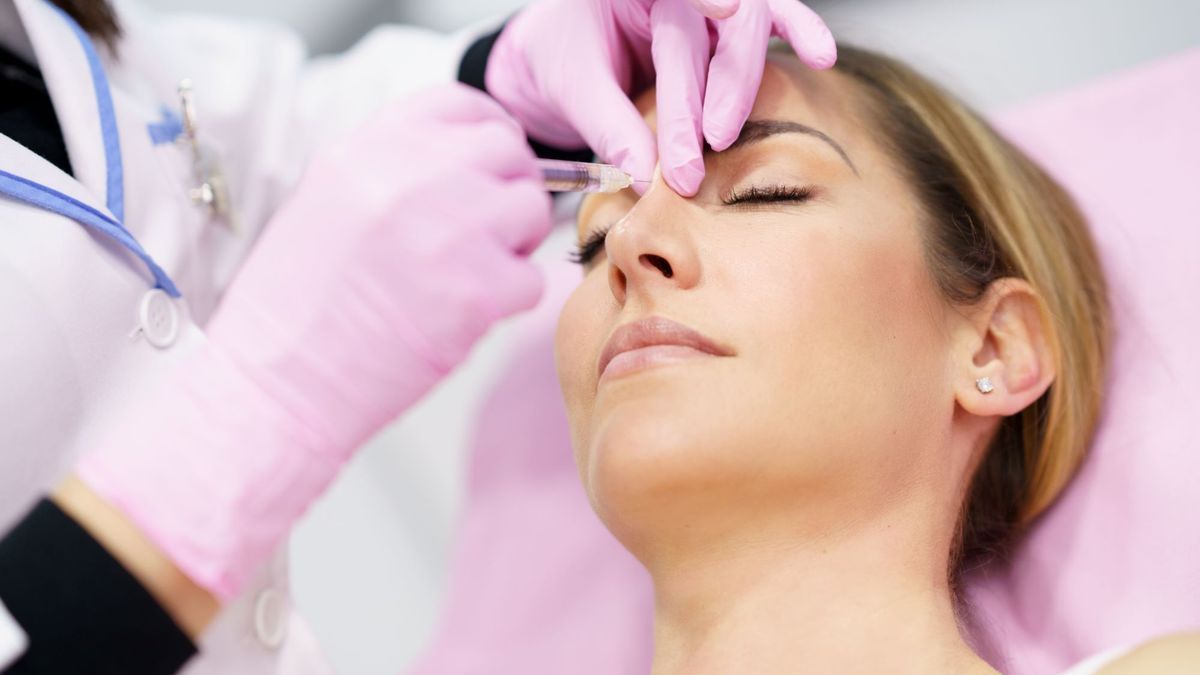
Only doctors are authorized to perform hyaluronic acid injections
The National Medicines Safety Agency reminds in this respect that injections carried out without authorization are dangerous and prohibited: “only doctors are authorized to perform them”.
Indeed, the success of an injection is based on several criteria: the hygienic conditions in which it is carried out (change of syringe, bottle, disinfection of premises, etc.); the choice of the injectable product depending on the injection area; traceability and good conservation of the injected product; the anatomy of the face and body; carrying out prior to an interrogation; analysis of the patient’s medical history (allergies, autoimmune diseases, etc.)
If not done correctly, hyaluronic acid injections can cause side effects, including allergic reactions, bleeding or subcutaneous infections.
Non-medical injections: what are the risks involved?
They are multiple and varied.
- An allergy to the injected product (anaphylactic shock);
- A local infection in the area where the product was injected (which can turn into septicemia if the infection is not treated in time);
- Viral (HIV, hepatitis C) or bacterial contamination if the equipment used has already been used;
- Necrosis or ischemia which may lead to amputation of tissues if the product is injected into a blood vessel;
- Loss of vision if the product is injected into a vessel that irrigates the eye.
In the long term, the risks are migration of hyaluronic acid and inflammation of the injected tissues.
In the event of adverse effects, the doctor can prescribe appropriate treatment. Hospitalization is sometimes necessary.
“Doctors know how to manage immediate adverse effects such as an allergy to the injected product – anaphylactic shock –, necrosis or ischemia – blocked blood vessel –, through appropriate medical treatment.concludes the ANSM.
A cry of warning from cosmetic surgeons
Although the European regulation recommends strict traceability of these products, this new decision does not go in the direction of the cry of warning from cosmetic surgeons. Last March, they published an article in Le Parisien, asking the authorities to ban the over-the-counter supply of hyaluronic acid.
For its part, in April, the Ministry of Health and Prevention announced that it had “initiated work to restrict access to injectable hyaluronic acid products to only healthcare professionals authorized to administer them and to people with a medical prescription“. While specifying that a “draft decree to this effect is currently being consulted with stakeholders in the sector“. To be continued, therefore.
