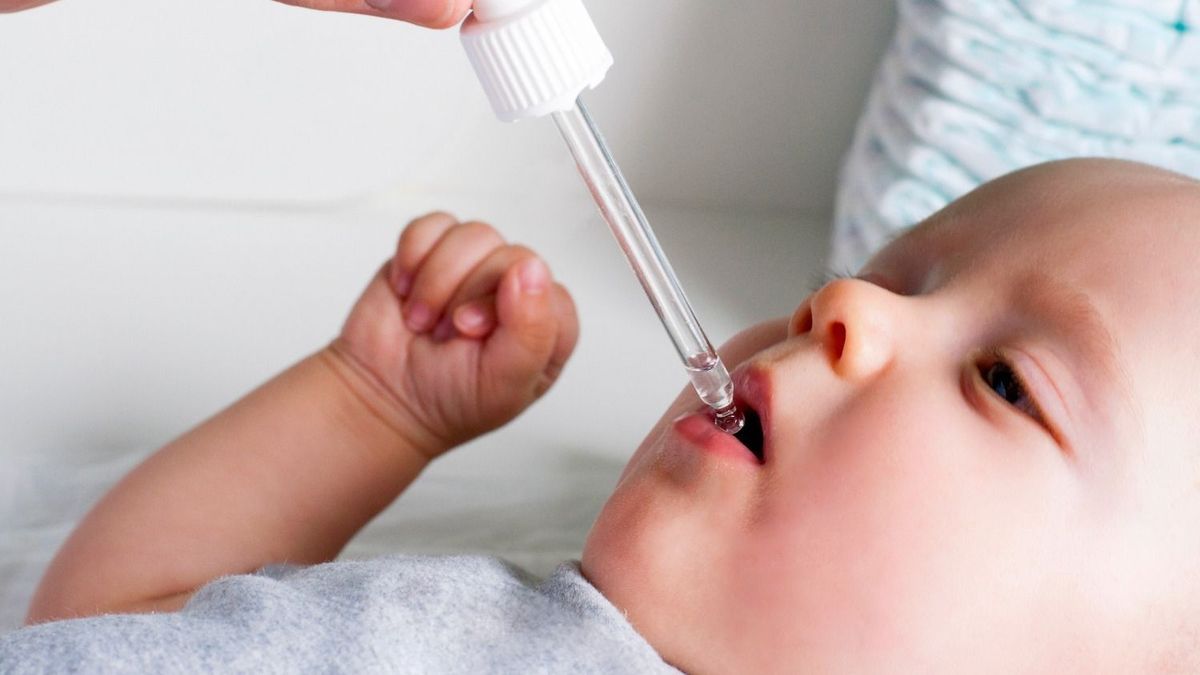
Is your baby anemic? Caution. The measuring pipette currently supplied with Ferrostrane 0.68% syrup could lead to a risk of overdose. Explanations.
The Ferrotrane pipette, commonly used in infants in cases of iron deficiency, should be handled with caution. According to the ANSM, it would be unsuitable for low weight infants.
Unsuitable graduations
According to the latest ANSM bulletin, the measuring pipette currently supplied with Ferrostrane 0.68% syrup and indicated in particular in low-weight babies for curative or preventive treatment of iron deficiency, should be handled with care. precaution.
She is indeed “unsuitable for low weight infants because its graduations do not allow very small volumes to be collected“, warns the ANSM.
The infant then risks ingesting iron in too large quantities, which can cause various side effects, more or less dangerous:
- Diarrhea;
- Constipation;
- Nausea ;
- Vomitings ;
- Digestive bleeding.
“In infants, these side effects can be particularly severe.“, warns the agency.
All healthcare professionals involved have been informed of these risks.
Ferrotrane: how to avoid an overdose?
To avoid any risk of medication error, particular attention must be paid to the prescribed volume, warns the ANSM.
“The latter can be between two graduations of the current pipette (0.7 mL/day or 1.3 mL/day for example), or even be less than 0.5 mL of the first graduation (0.3 mL/day)“, she explains.
The good news ? The risk of overdose should soon decrease.
During the second half of 2024, a pipette suitable for small volumes will in fact be delivered with the syrup, in new packaging.
Two types of Ferrotrane 0.68% containing the same syrup will thus be available, details the ANSM:
- One with the current pipette graduated from 0.5 to 0.5 mL, starting at 0.5 mL, for most patients and the majority of infants;
- The other with a pipette with finer graduations and adapted to the small volumes which can be prescribed for low weight babies.

